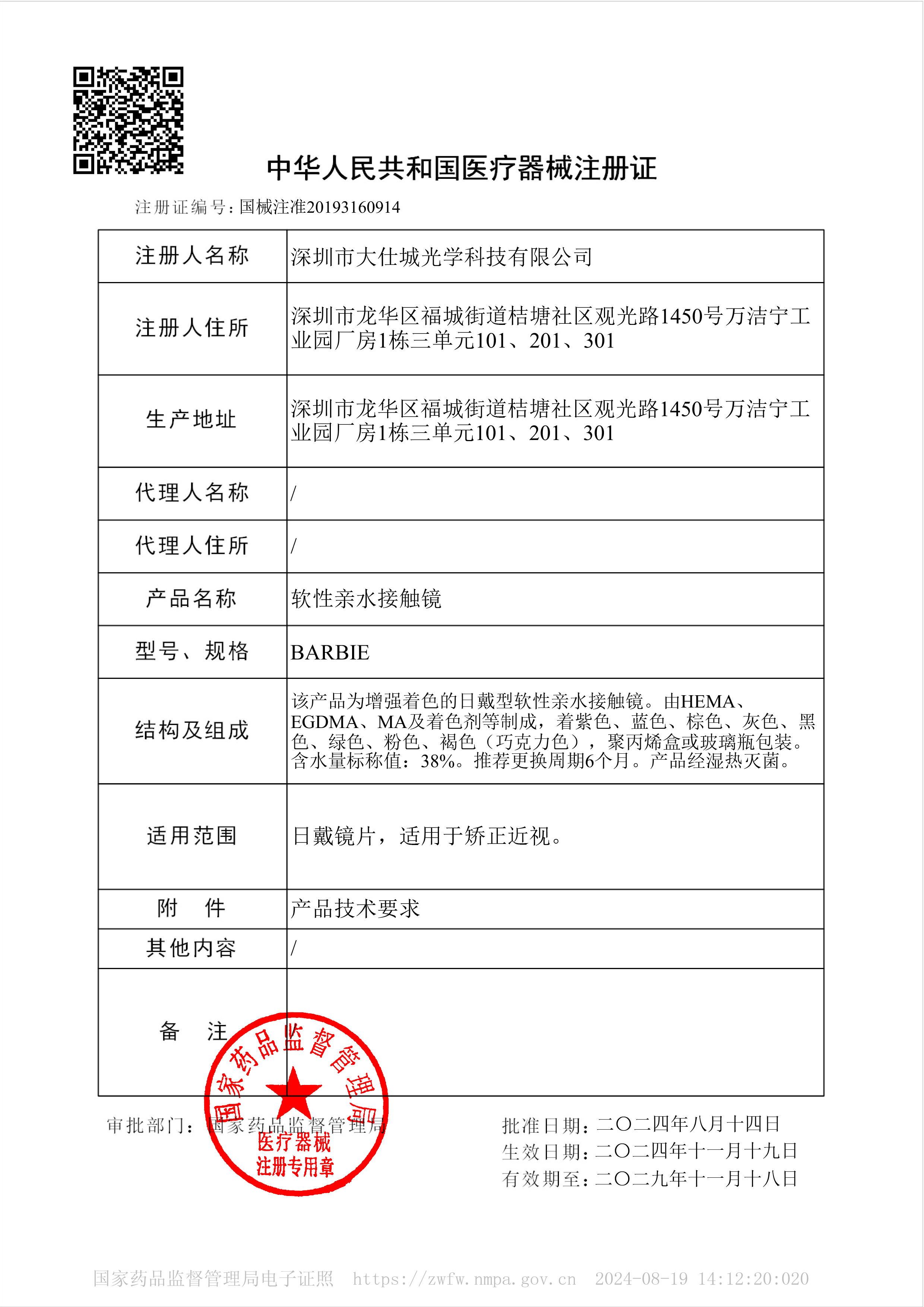
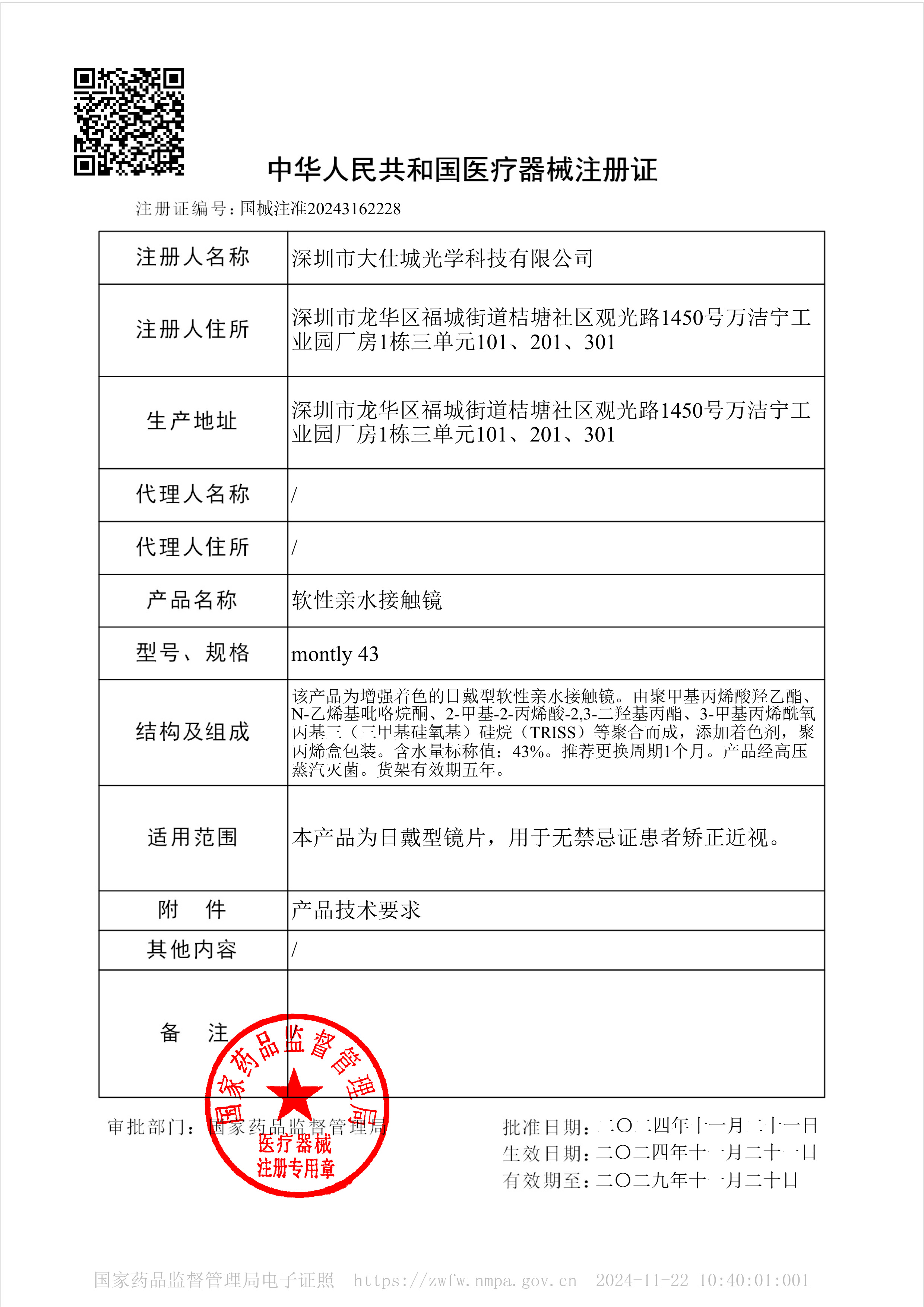
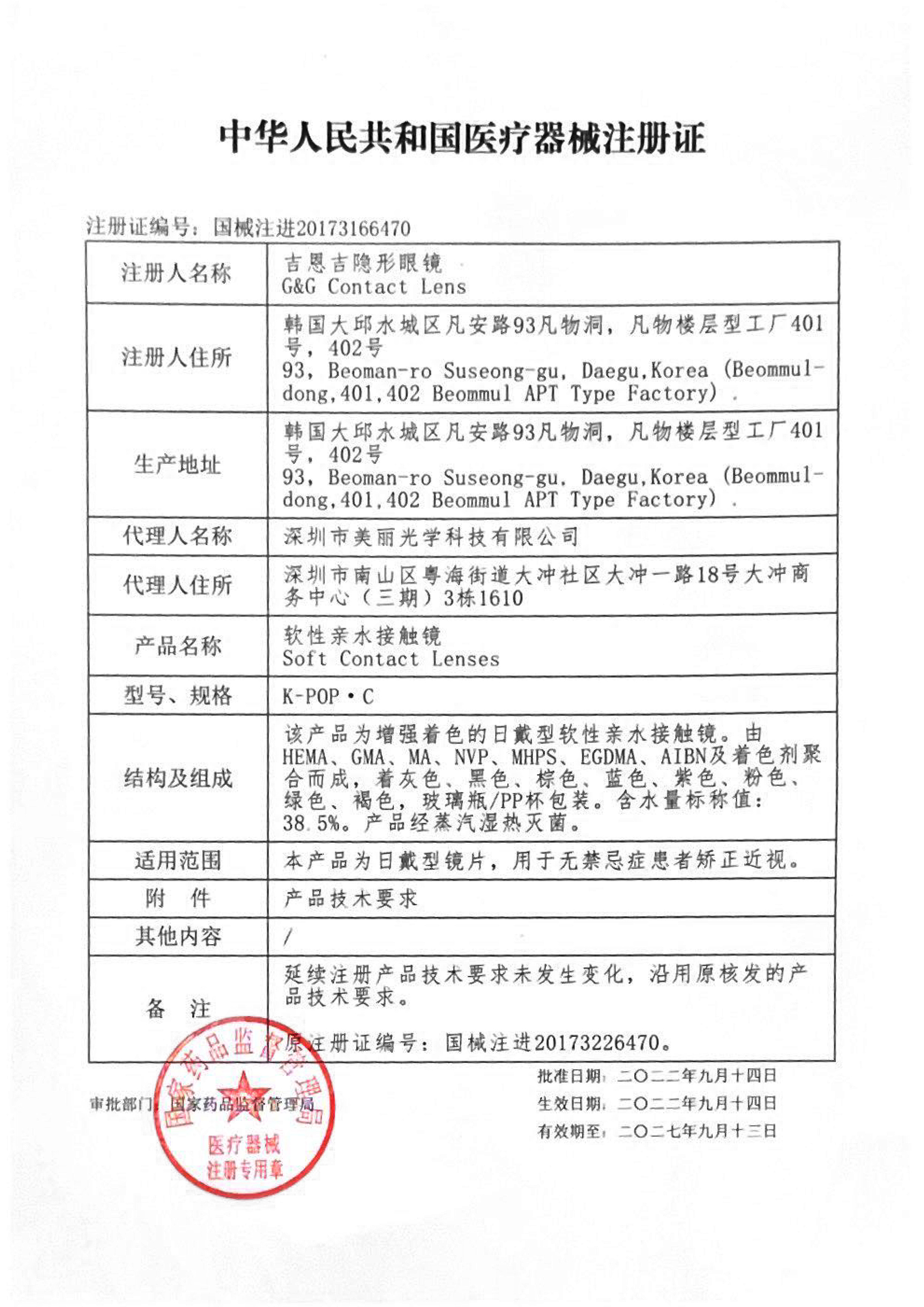
According to official FDA data as of September 2025, a mere 62 manufacturers globally hold marketing authorization for soft contact lenses, a Class II medical device. This designation demands strict adherence to protocols, including 51(k) clearance, sterility and bio-compatibility testing, and UDI labeling compliance. Establishing standards as the first and only manufacturer from mainland China to join this elite global cohort. This FDA certification serves as a pivotal cornerstone for our global expansion, empowering our entry into premium American and European markets, while concurrently advancing regulatory compliance across Southeast Asia, the Middle East, and beyond.